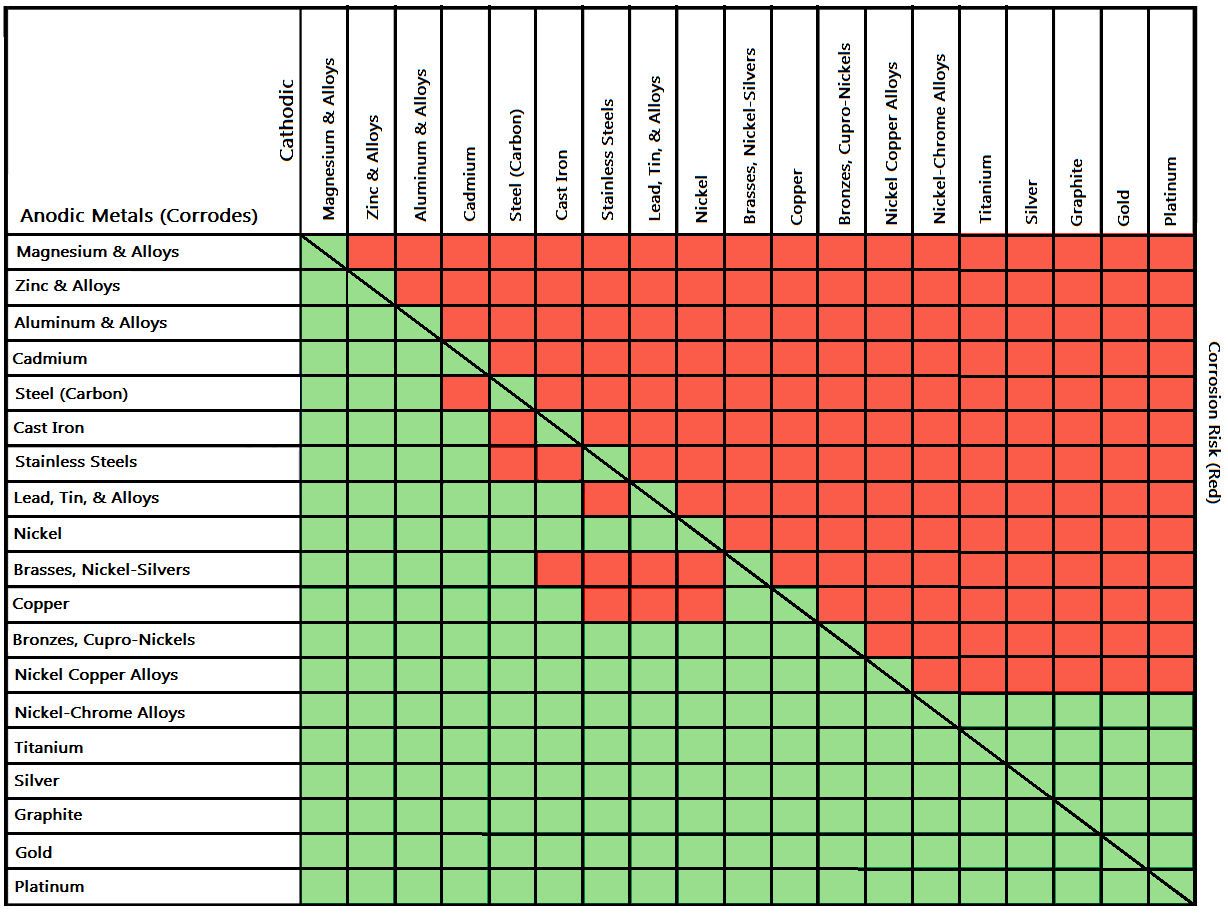
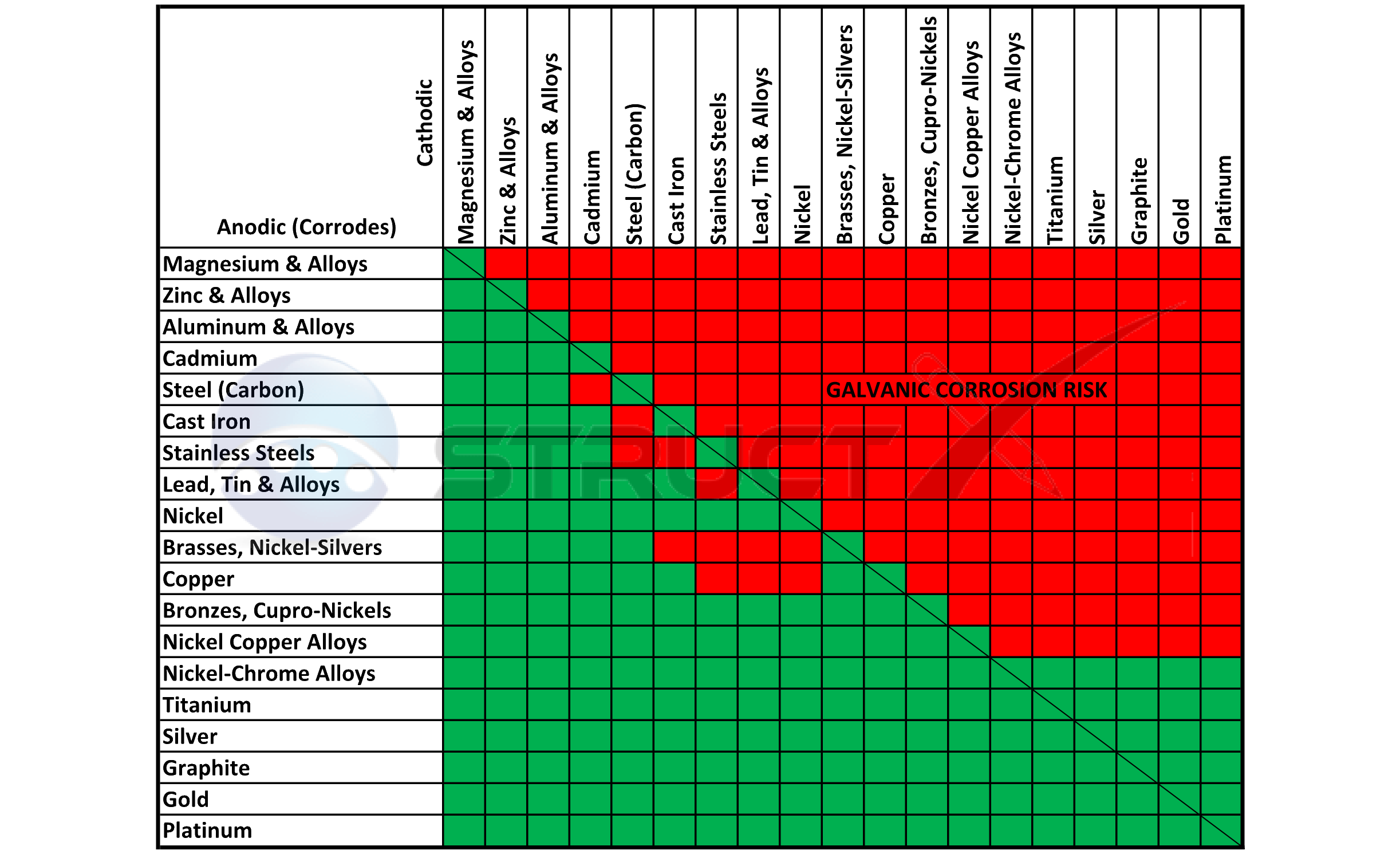
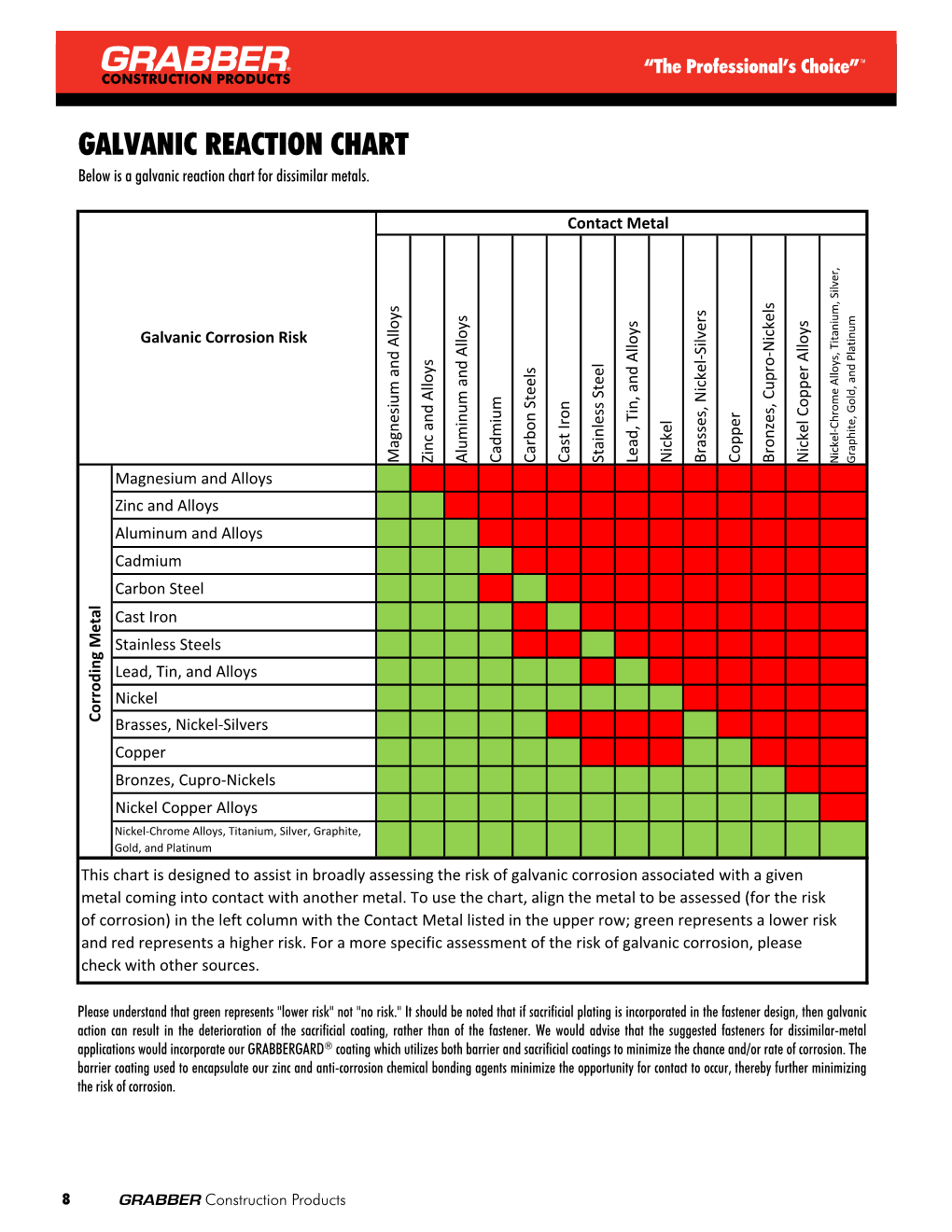
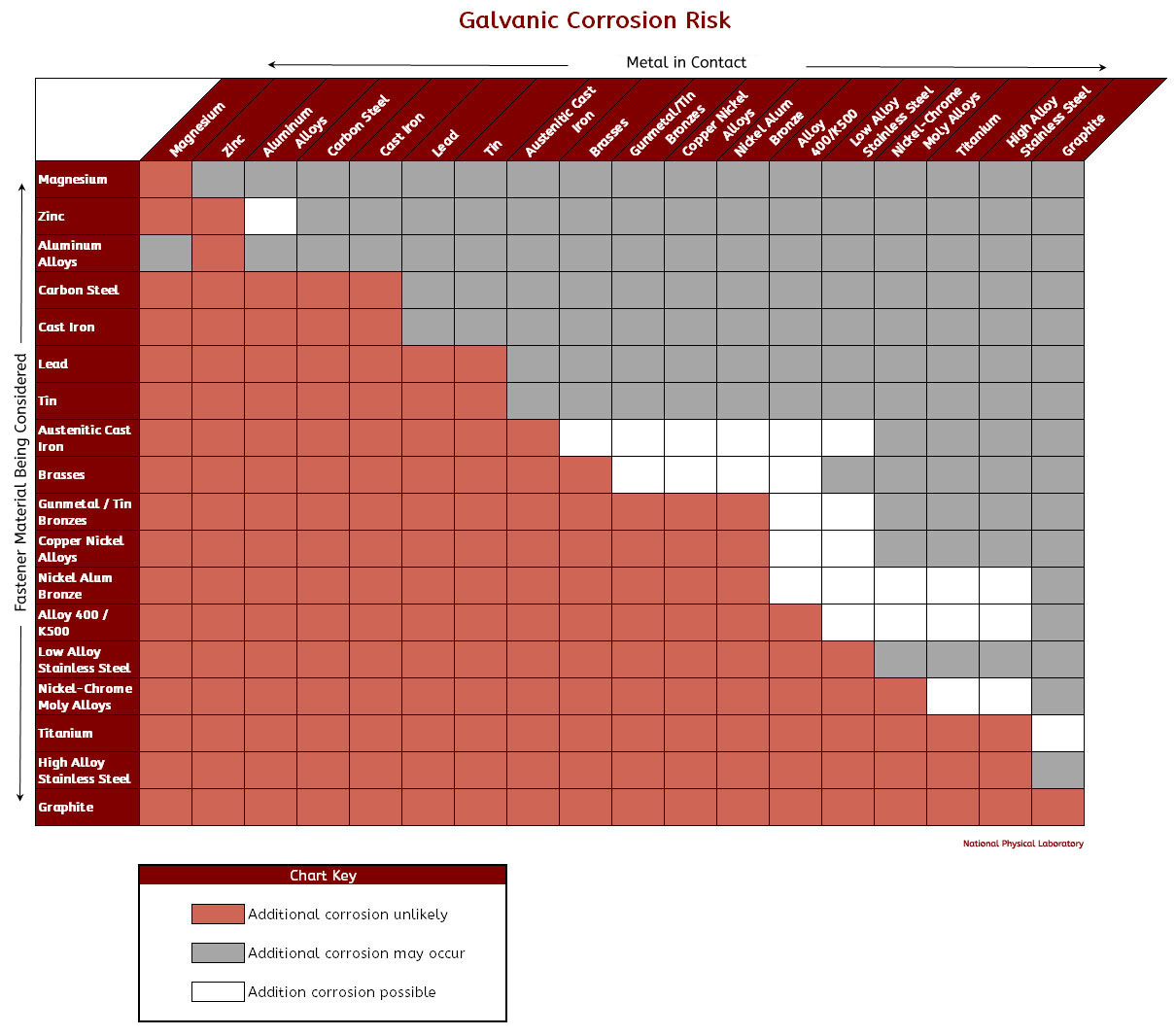
Galvanic Action Chart
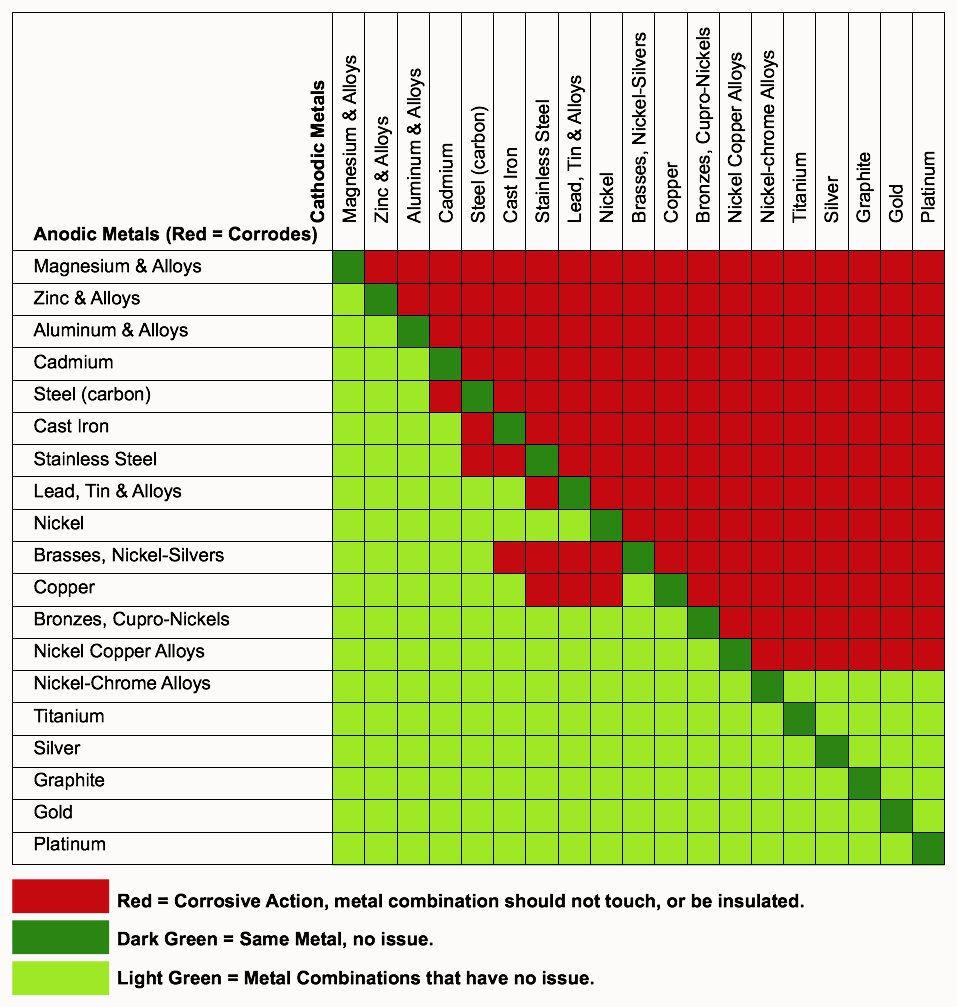
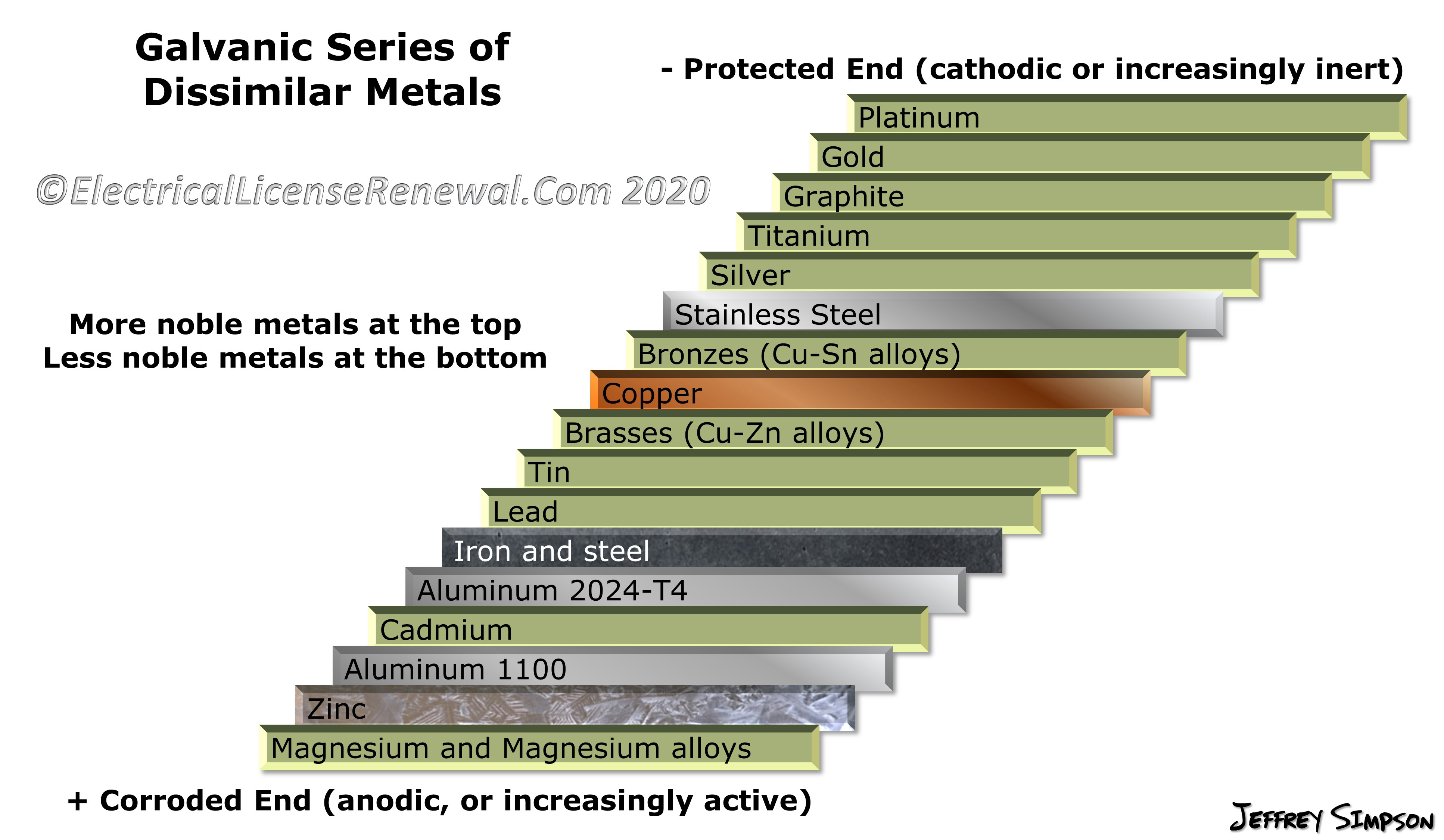
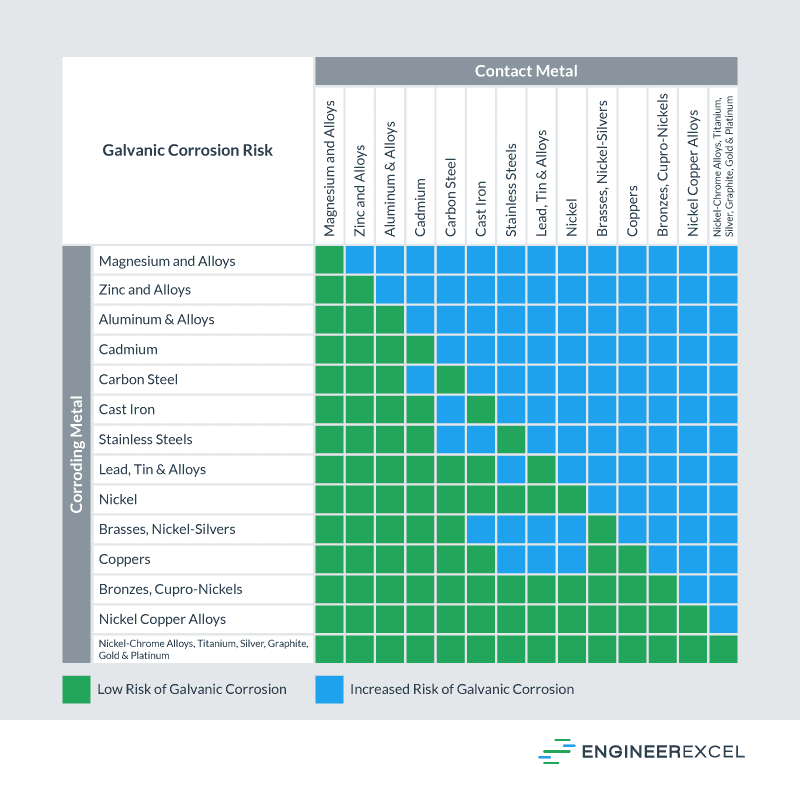
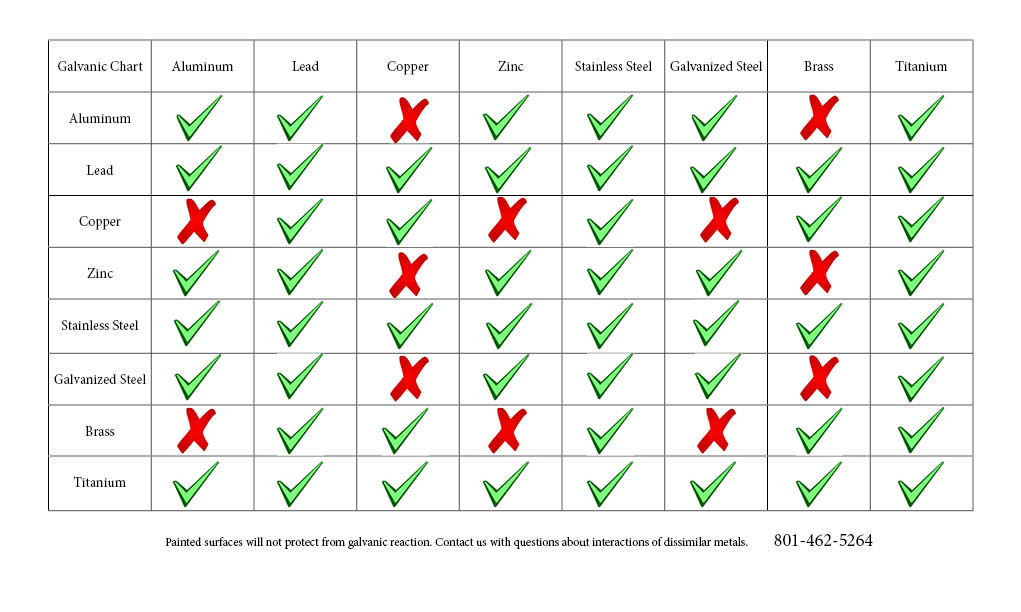
Galvanic Action Chart - Though the order of metals in a. Web below is a galvanic reaction chart for dissimilar metals. The closer together the material are on the chart to the right, the less. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amounts of. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical. The list begins with the more. See a chart of metals and their electrochemical potentials, and examples. Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. Web below is the galvanic corrosion chart, along with steps for using the chart properly to determine metal compatibility. In this article, we'll look at an example to illustrate the use. Web below is a galvanic reaction chart for dissimilar metals. The list begins with the more. Web learn what galvanic action is, how it occurs, and how to prevent it in construction. The below galvanic corrion chart or anodic index table shows anodic index for different materials. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. Web there are four elements necessary for corrosion to occur in a galvanic cell: This can help you in the selection of the best. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. We consider two metals compatible if their emf. The below galvanic corrion chart or anodic index table shows anodic index for different materials. Web the galvanic corrosion table ranks metals from the most “active” to the least active. Zinc coatings on carbon steel and. Web the galvanic corrosion chart, also referred to as the galvanic series or compatibility chart, is a valuable tool used to assess the compatibility. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amounts of. Web galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or corrodes. Web to minimize galvanic corrosion, select fasteners based on their. Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. Web the galvanic corrosion table ranks metals from the most “active” to the least active. The closer together the material are on the chart to the right, the less. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an. Web the galvanic corrosion table ranks metals from the most “active” to the least active. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. Web galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. See a chart of metals and their electrochemical potentials, and examples. Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. Web view this chart of galvanic compatibility. Web electrolytic corrosion (electrolysis) occurs. The most active metals in the galvanic corrosion chart, like aluminum, zinc, or. Web below is a galvanic reaction chart for dissimilar metals. Web to minimize galvanic corrosion, select fasteners based on their material compatibility with the substrates. The closer together the material are on the chart to the right, the less. Web however, you can completely avoid galvanic corrosion. So, for example, choosing zinc on zinc would have the lowest risk for. Web below is the galvanic corrosion chart, along with steps for using the chart properly to determine metal compatibility. The list begins with the more. We consider two metals compatible if their emf. The below galvanic corrion chart or anodic index table shows anodic index for different. In this article, we'll look at an example to illustrate the use. The below galvanic corrion chart or anodic index table shows anodic index for different materials. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amounts of. The most active metals in the galvanic. Web view this chart of galvanic compatibility. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web there are four elements necessary for corrosion to occur in a galvanic cell: Web the galvanic corrosion table ranks metals from the most “active” to the least active. Web below, we give. Web below is a galvanic reaction chart for dissimilar metals. The following galvanic table lists metals in the order of their relative activity in seawater environment. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence. Web there are four elements necessary for corrosion to occur in a galvanic cell: Though the order of metals in a. Web the galvanic corrosion table ranks metals from the most “active” to the least active. The closer together the material are on the chart to the right, the less. See a chart of metals and their electrochemical potentials, and examples. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amounts of. We consider two metals compatible if their emf. Web below is the galvanic corrosion chart, along with steps for using the chart properly to determine metal compatibility. The following galvanic table lists metals in the order of their relative activity in seawater environment. Web a chart depicting the galvanic series for some common metals in a frequently encountered conducting solution, seawater, is included in figure 1. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical. This can help you in the selection of the best. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. The below galvanic corrion chart or anodic index table shows anodic index for different materials. Web view this chart of galvanic compatibility.Galvanic Corrosion Common Questions Answered
Galvanic Series (electrochemical series)
Galvanic Reaction Chart
Galvanic Corrosion Chart
Galvanic Action Corrosion Prevention Architect's Blog
Galvanic Action Chart
Galvanic Reaction Chart All Points Fasteners
Galvanic Corrosion Between Metals Chart
Galvanic Action Chart PDF
Galvanic Chart For Metals
Web However, You Can Completely Avoid Galvanic Corrosion By Choosing Matching Metal Anchors.
So, For Example, Choosing Zinc On Zinc Would Have The Lowest Risk For.
The List Begins With The More.
Web Galvanic Corrosion (Some Times Called Dissimilar Metal Corrosion) Is The Process By Which The Materials In Contact With Each Other Oxidizes Or Corrodes.
Related Post: