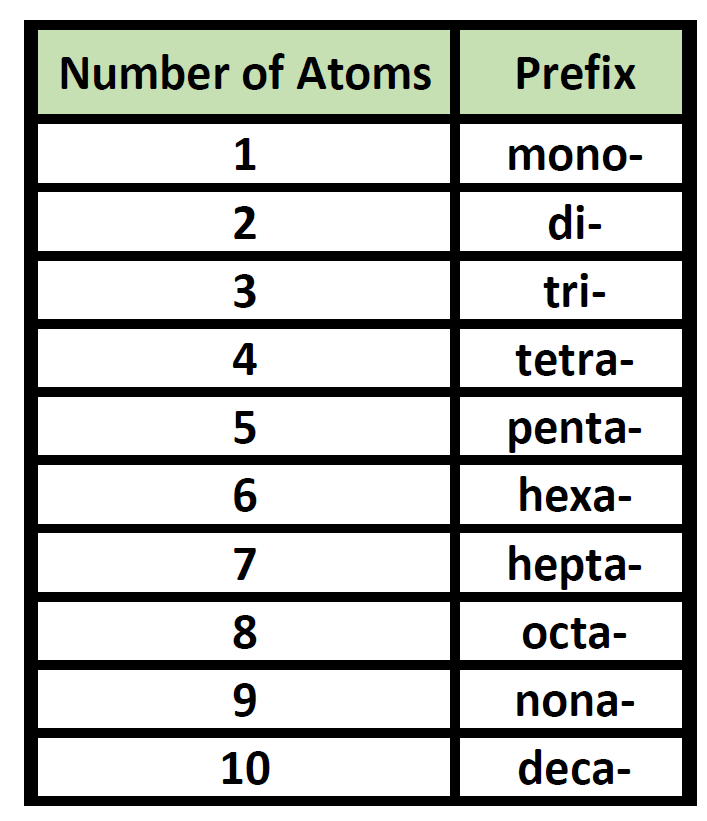
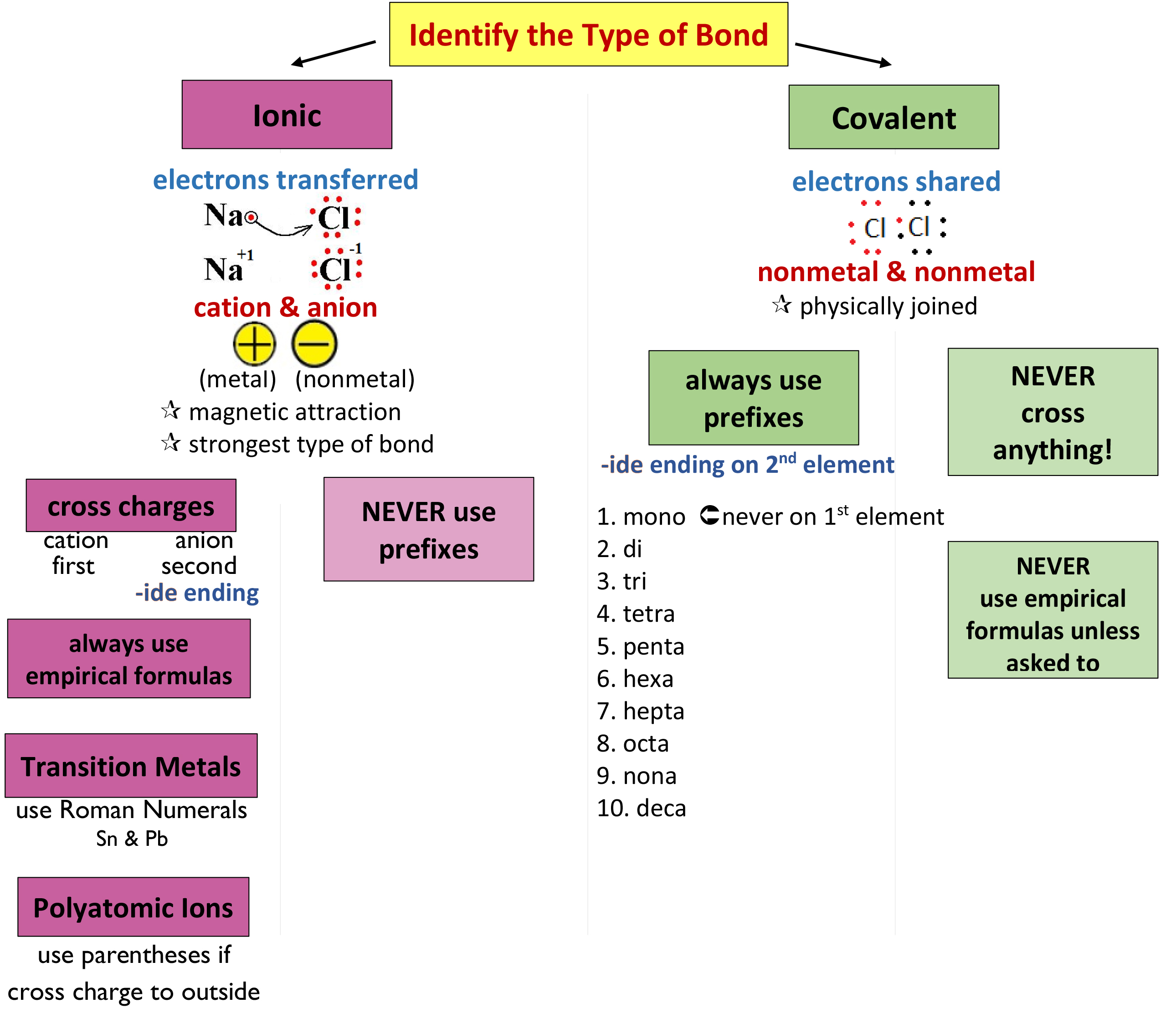
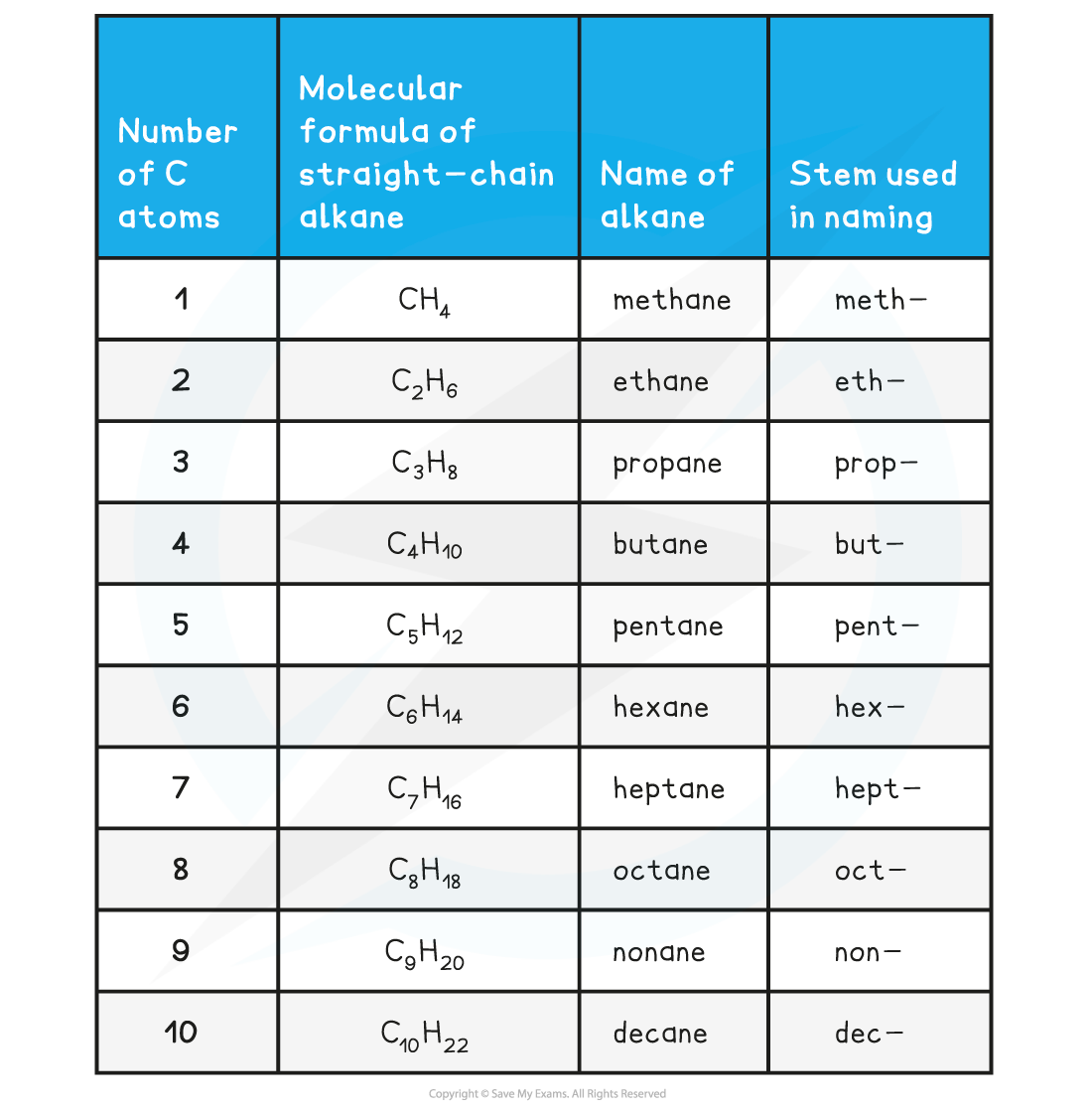
Chemistry Nomenclature Chart
Chemistry Nomenclature Chart - The basics of organic nomenclature, of inorganic nomenclature and polymer nomenclature are summarized in a collection of brief guides accessible below. And so, iupac (think of the “ministry of magic”, but for chemists) has developed one. It is published in the nomenclature of organic chemistry (informally called the blue book ). Web we need some kind of priority system for nomenclature. There are several general rules, however, that can bring some order out of this apparent chaos. Chemistry of the main groups and transition elements, including typical. Web table 2.7.1 2.7. Structure and stereoisomerism, importance of coordination compounds (in qualitative analysis, extraction of metals and. Web derive names for common types of inorganic compounds using a systematic approach. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and n 2 o 4. Chemistry of the main groups and transition elements, including typical. Web chemical nomenclature for acids. For instance, hcl is a common. Web nomenclature, a collection of rules for naming things, is important in science and in many other situations. There does not exist any particular collection of rules for writing the trivial naming of compounds. Web in chemical nomenclature, the iupac nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended [1] [2] by the international union of pure and applied chemistry (iupac). This chapter describes an approach that is used to name simple ionic and molecular compounds, such as nacl nacl, caco3 caco 3, and n2o4 n 2 o 4. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and n 2 o 4. The following areas are normally included on the exam: Common greek prefixes used in naming simple moleculs. Web in chemical nomenclature, the iupac nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended [1] [2] by the international union of pure and applied chemistry (iupac). There are some rules we need to follow,which are: This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco. Web in chemical nomenclature, the iupac nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended [1] [2] by the international union of pure and applied chemistry (iupac). Common greek prefixes used in naming simple moleculs. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3,. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. The nomenclature used most frequently worldwide is the one created and developed by the international union of pure and applied chemistry (iupac). Names of organic compounds are simplified via this system. If you have a molecule with, say, a carboxylic acid and a. Because the elements are chemically combined, they cannot be separated using physical means. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. These compilations are coordinated by the division of chemical nomenclature and structure representation. Common greek prefixes used in naming simple moleculs. There does not exist any particular collection of rules. The nomenclature used most frequently worldwide is the one created and developed by the international union of pure and applied chemistry (iupac). Chemical nomenclature is far too big a topic to treat. Names of organic compounds are simplified via this system. Web chemical nomenclature for acids. Web at first glance, the nomenclature of the polyatomic negative ions in the table. Common greek prefixes used in naming simple moleculs. There does not exist any particular collection of rules for writing the trivial naming of compounds. The following areas are normally included on the exam: Web chemical nomenclature for acids. Chemical reactivity and products of chemical reactions; Common greek prefixes used in naming simple moleculs. He functional group with the highest priority will be the one which gives its suffix to the name of the. Chemical nomenclature is a set of rules to generate systematic names for chemical compounds. Acid usually means an anion combined with the hydrogen ion as the cation. This module describes an approach. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and n 2 o 4. A compound is a substance that contains two or more elements chemically combined in a fixed proportion. There. There are some rules we need to follow,which are: There are several general rules, however, that can bring some order out of this apparent chaos. Chemical nomenclature is a set of rules to generate systematic names for chemical compounds. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and. Web iupac provides recommendations on many aspects of nomenclature. Chemical nomenclature is a set of rules to generate systematic names for chemical compounds. There does not exist any particular collection of rules for writing the trivial naming of compounds. Chemistry of the main groups and transition elements, including typical. Nomenclature, a collection of rules for naming things, is important in. This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and n 2 o 4. As one of its major activities, iupac. Chemical reactivity and products of chemical reactions; There are some rules we need to follow,which are: These compilations are coordinated by the division of chemical nomenclature and structure representation. He functional group with the highest priority will be the one which gives its suffix to the name of the. The nomenclature used most frequently worldwide is the one created and developed by the international union of pure and applied chemistry (iupac). Iupac nomenclature ensures that each compound (and its various isomers). This module describes an approach that is used to name simple ionic and molecular compounds, such as nacl, caco 3, and n 2 o 4. Nomenclature, a collection of rules for naming things, is important in science and in many other situations. Bonding, werner’s theory, vbt, and cft; And so, iupac (think of the “ministry of magic”, but for chemists) has developed one. Introduction to chemical nomenclature is shared under a cc by 3.0 license and was authored, remixed, and/or curated by stephen lower via source content that was edited to the style and standards of the libretexts platform. Names of organic compounds are simplified via this system. Chemistry of the main groups and transition elements, including typical. If there are two atoms, place the more metallic first (furthest to the left on the periodic table) is there is a central atom, place it first, and the atoms attached after.Organic Chemistry Nomenclature Cheat Sheet Cheat Sheet
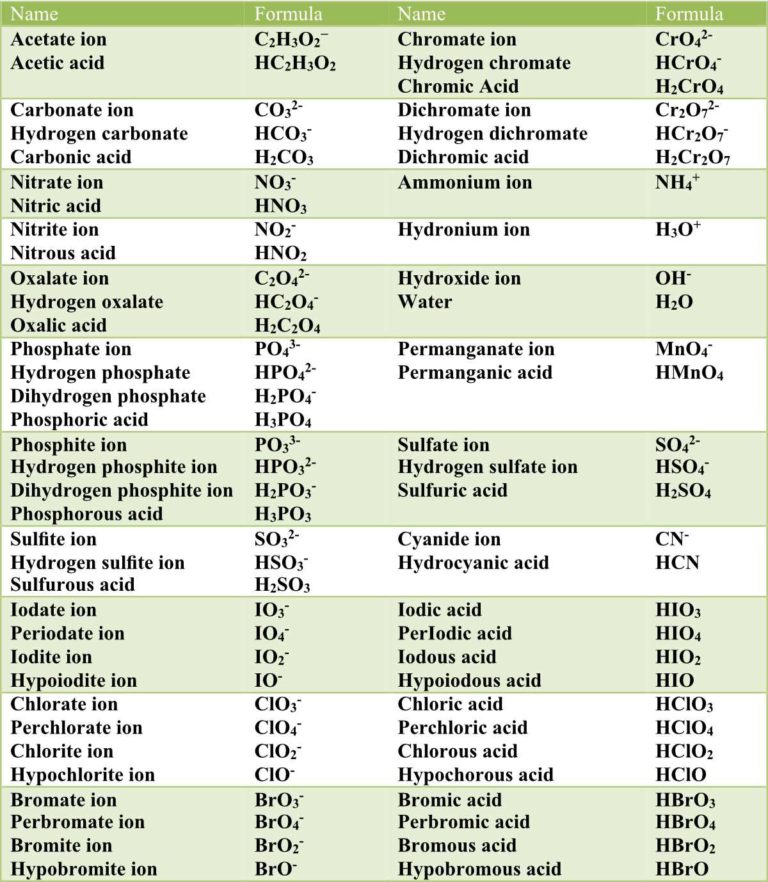
Nomenclature of Acids Pathways to Chemistry
NEET Chemistry Nomenclature of Organic Compounds IUPAC Rules YouTube
Chemistry Chart
Chemistry Nomenclature Chart
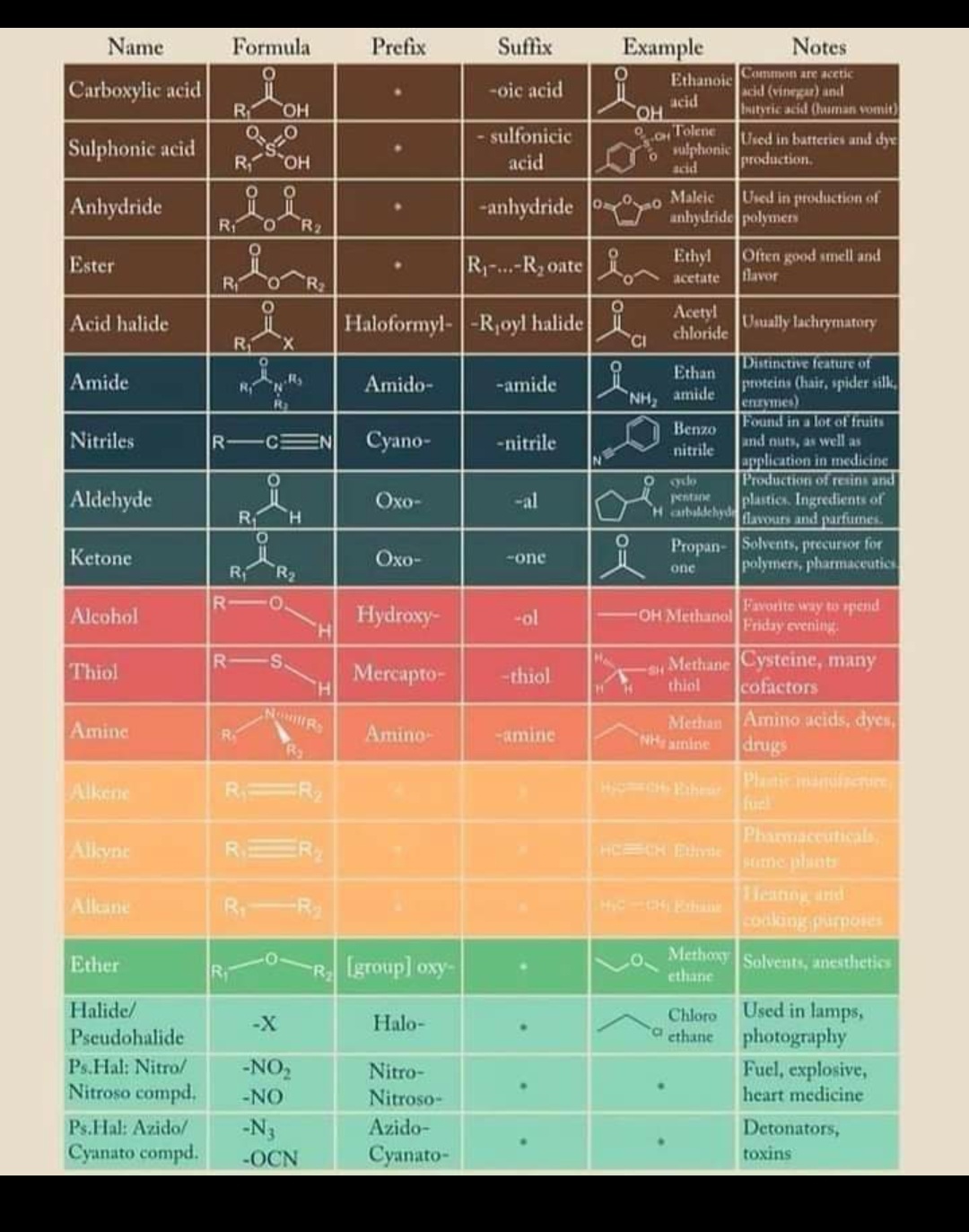
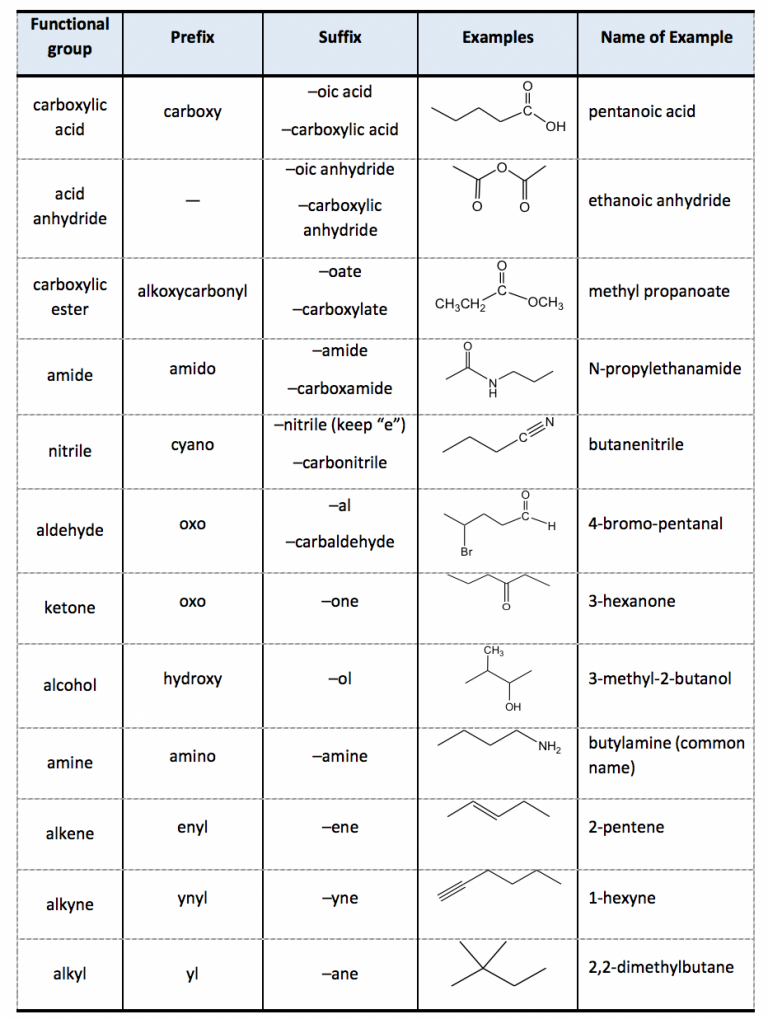
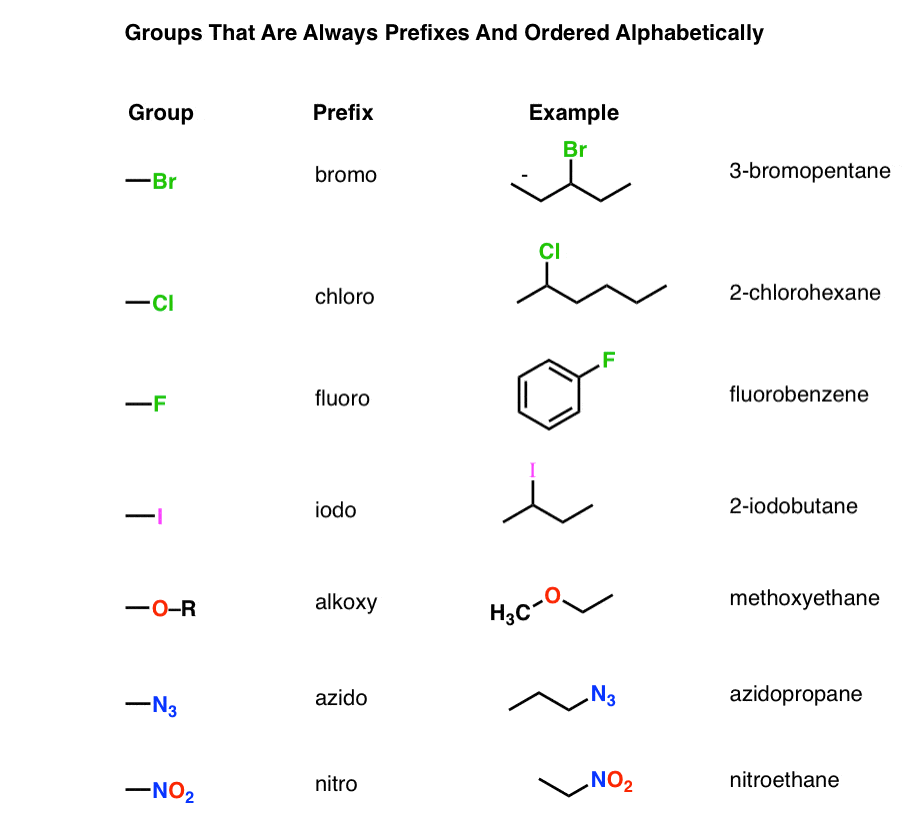
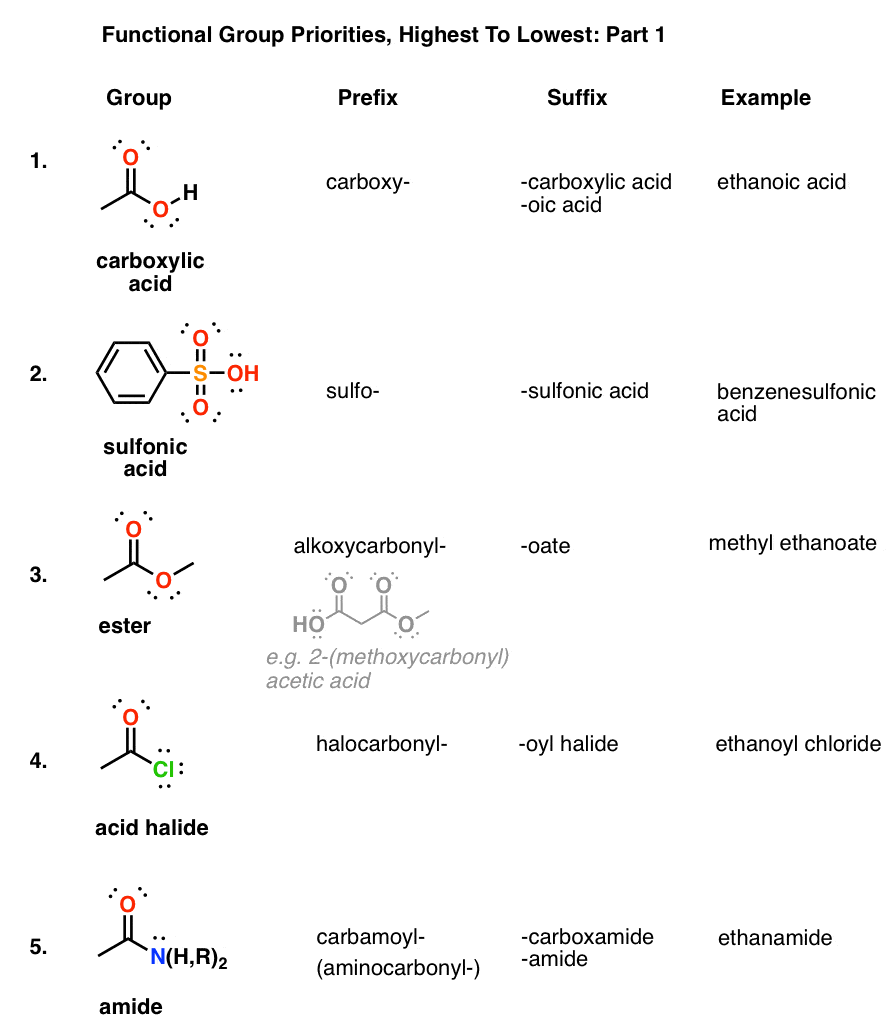
Table of Functional Group Priorities for Nomenclature Master Organic
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
07 Nomenclature Mrs. Cook's Chemistry Class
Table of Functional Group Priorities for Nomenclature Master Organic
IB DP Chemistry HL复习笔记10.1.3 Nomenclature翰林国际教育
The Basics Of Organic Nomenclature, Of Inorganic Nomenclature And Polymer Nomenclature Are Summarized In A Collection Of Brief Guides Accessible Below.
In Addition, The Properties Of The Compound (Both Physical And Chemical) Are Radically.
Web Table 2.7.1 2.7.
Examples Of This Include Phenol, Acetic Acid, And Toluene.
Related Post: